Traceability solution for cartons and cases in a blister line
22 February 2018
HYDRA
23 March 2018X-RAY INSPECTION FOR THE PHARMACEUTICAL INDUSTRY WHEN VISUAL INSPECTION CANNOT WORK
The Soft X-ray system has purposely been developed for the pharma packaging industry; the lower permeability of these rays, in fact, makes it possible to increase the contrast.Thanks to this characteristic not only the product but also the package as well as the accessories can be analyzed.
The X-ray inspection represents the best solution to ensure the integrity of the sealed product and to prevent risks, such as customer complaints and market recalls.
The X-ray system determines the density of the individual visible points, in order to make the single product, its weight and its integrity inside the package, visible and recognizable.
A single system is therefore able to perform a complete product inspection, otherwise impracticable:
- Product faults such as deformations, lacks, breakages or contaminations
- Incomplete packages containing more accessories, such as:
– Blisters with pre-filled syringes and needle
– Flow packs containing products and leaflets
– Strips containing capsules or tablets
– Cartons containing canister with inhaler and leaflet
- Product or package contamination, such as: glass, metals, etc…
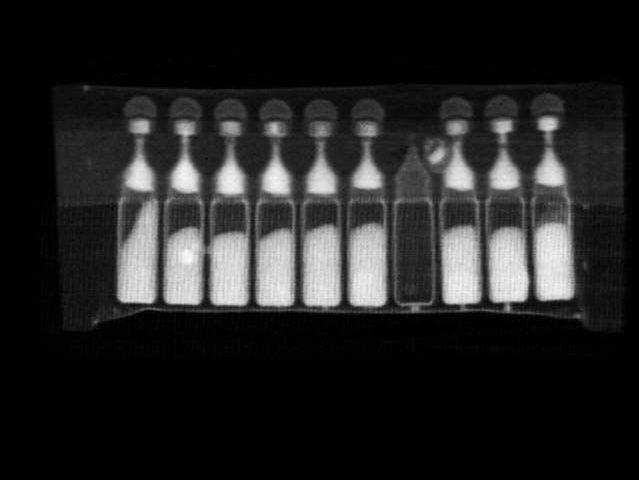
- Filling faults of bottles, vials, capsules, syringes and single-doses
- Dynamic weighing, including the single capsule or product, and detection of air bubbles.
The device is easy to set up in order to make use of the features mentioned above in a very intuitive and customizable manner.
The low kinetic energy (<30Kev) and the low radiation dose do not include the machine in the X-ray standard (Art. 8 Annex I of the Legislative Decree 230/1995 and subsequent amendments and additions), therefore, considering the low kinetic energy of these X-rays, no periodic certification is required for the operator’s safety.
- Soft gel pills in blister—one product is empty
- Single-dose strip in flowpack—one product is empty
- Suppositories in strip—one product is not complete
- Vials in tray—one vial is broken
- Tablets in strip—one tablet is broken